With new developments in catalysis research and technology, catalysts are becoming increasingly important across the globe. They can be used to select specific products from a reaction.Ĭatalysts play an essential role in modern chemistry, allowing us to carry out complex chemical reactions quickly, cheaply and with minimal environmental impact.They can reduce the amount of waste produced from a reaction.They can be used to increase the yield of products from a reaction.They allow reactions to take place at lower temperatures, saving energy and reducing costs.They are also used in automobile engines and pollution control systems, as well as in medical treatments such as cancer therapies. Uses of CatalystsĬatalysts have a wide range of applications, including industrial processes such as the production of fertilizers, petrochemicals, pharmaceuticals and fine chemicals. Examples of homogenous catalysts include enzymes, acids and bases, while heterogeneous catalysts can include metals, zeolites and clays. Homogenous catalysts are those that exist in the same phase as the reactants, while heterogeneous catalysts are found in different phases. 
Types of CatalystsĬatalysts can be divided into two main categories: homogeneous and heterogeneous. 
The first use of a catalyst in a commercial application dates back to 1836 when platinum was used to speed up the production of sulfuric acid. History of CatalysisĬatalysis has been studied for centuries, with ancient alchemists first experimenting with catalysts to alter the properties of chemicals. Catalysts are becoming increasingly important across the globe. It is an agent which helps bring about a change without itself undergoing any permanent alteration. What is a Catalyst?Ī catalyst is a substance which accelerates a chemical reaction without being consumed or altered in the process. The use of catalysts has been documented since antiquity and is only becoming more important as we move towards a more sustainable future. There are two main types of catalysts - homogeneous and heterogeneous - each with their own advantages and disadvantages. ConclusionĬatalysts play an integral role in many industrial processes, from producing fuels and plastics to cleaning up pollution. Additionally, catalysts are being used to develop new materials for construction and electronics, as well as novel ways to recycle waste materials and reduce pollution. Renewable energy sources, such as wind and solar power, require the use of catalysts for efficient energy conversion. The Future of CatalystsĬatalysts are becoming even more important as we move towards a more sustainable future. Since then, catalysts have been used in a variety of industries, from automotive manufacturing to food production, helping to revolutionise many sectors. In the 19th century, chemists began to explore how catalysts worked and discovered that they had a major impact on the speed and efficiency of chemical reactions. 
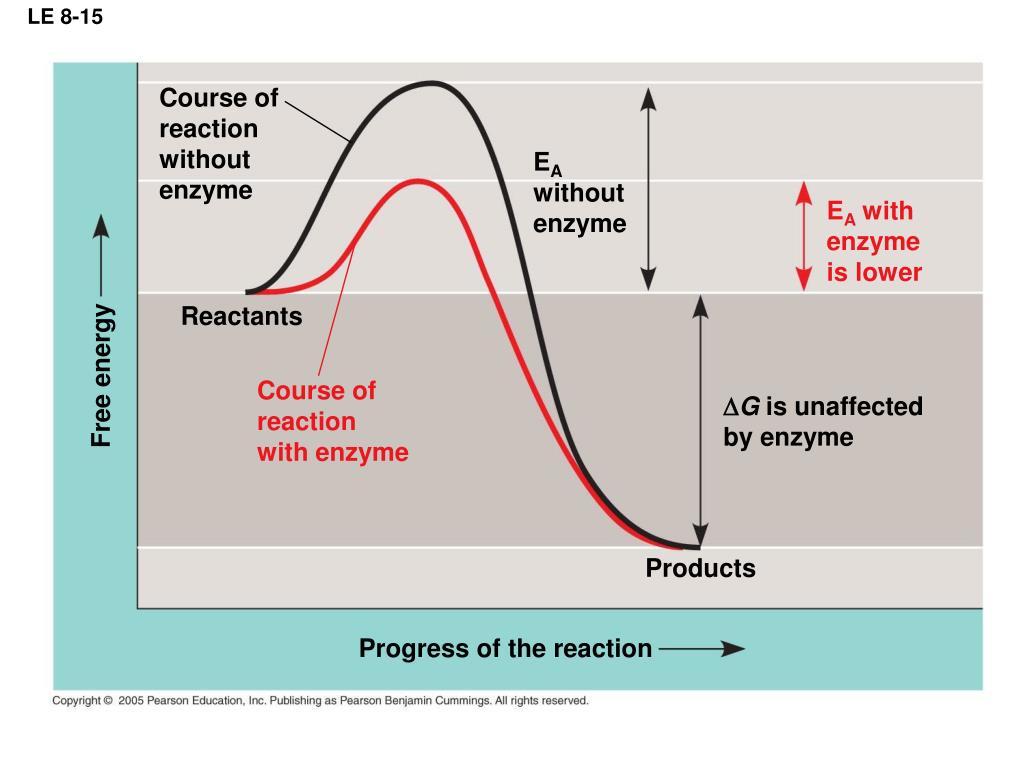
The use of catalysts has been documented since antiquity, with the earliest known use dating back to ancient Egypt. Both types of catalysts can be used in different types of reactions and have different advantages and disadvantages. Heterogeneous catalysts, on the other hand, are solid particles that are not dissolved in the reactants, such as carbon or zeolites. Homogeneous catalysts are those that are dissolved in the reactants themselves and are typically made from metals such as platinum or palladium. There are two main types of catalysts: homogeneous and heterogeneous. Catalysts can also be used to clean up polluted air and water, as well as to reduce emissions from cars and other vehicles. This means that catalysts play an important role in many industrial processes, such as the production of fertilisers, plastics, pharmaceuticals and fuels. What Do Catalysts Do?Ĭatalysts work by lowering the amount of energy required for a reaction to take place, meaning that these reactions can be completed more quickly and efficiently than they could without the presence of a catalyst. As such, catalysts are becoming increasingly important across the globe. These substances allow for a wide range of chemical processes, from refining crude oil into fuel to converting food waste into energy, to take place safely and efficiently. Catalysts are substances that accelerate the rate of chemical reactions, allowing them to occur under milder conditions or with reduced energy input.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
